Tenvir Tablets
MOQ : 100 Packs
Tenvir Tablets Specification
- Molecular Formula
- C9H14N5O4P
- Usage
- Used in the treatment of HIV infection
- Salt Composition
- Tenofovir Disoproxil Fumarate
- Grade
- Pharmaceutical Grade
- Indication
- HIV-1 infection, chronic hepatitis B
- Pacakaging (Quantity Per Box)
- 30 Tablets per box
- Dosage Form
- Tablet
- Dosage
- 300 mg
- Assay
- Not less than 98%
- Origin of Medicine
- India
- Expiration Date
- 24-36 months from manufacturing date
- CAS No
- 147127-20-6
- Appearance
- White to off-white tablets
- Storage
- Store below 30C, protect from moisture
- Medicine Type
- Allopathic
- Suitable for Adults
- Yes
- Prescription Required
- Yes
- Shelf Life
- 2 to 3 years
- Controlled Substance
- No
- Packaging Type
- Bottle
- Route of Administration
- Oral
- Brand Name
- Tenvir
- Side Effects
- Nausea, headache, abdominal pain, dizziness
- ATC Code
- J05AF07
Tenvir Tablets Trade Information
- Minimum Order Quantity
- 100 Packs
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 5000 Packs Per Month
- Delivery Time
- 2-10 Days
- Main Domestic Market
- All India
About Tenvir Tablets
Discover the exquisite efficacy of Tenvir Tablets-now available at a reduced price during our exclusive ephemeral price cut! Manufactured in India and supplied in enviable bottle packaging of 30 tablets per box, Tenvir's pharmaceutical-grade formulation (Tenofovir Disoproxil Fumarate) offers unbeatable reliability for adults with HIV-1 infection and chronic hepatitis B. With an extensive shelf life of 2 to 3 years, Tenvir Tablets must be prescribed by a practitioner. The allopathic, non-controlled substance ensures an assay of not less than 98%. Seize this opportunity for an exceptional treatment option at an irresistible and limited-time offer.
Commercial Application and Suitability of Tenvir Tablets
Tenvir Tablets are primarily indicated for the treatment of HIV-1 infection and chronic hepatitis B, making them highly valuable in clinical and hospital settings. Suitable for adult patients, these allopathic tablets may also be used in combination regimens depending on practitioner advice. Their pharmaceutical grade ensures reliable results, while the well-established safety profile allows wide applicability where prescription medication for viral infections like HIV is required.
Export Market Reach and Shipping Solutions for Tenvir Tablets
Tenvir Tablets have secured a formidable presence across global export markets due to their quality and unbeatable price point. Supplying from India, logistics are streamlined to provide express shipping and efficient delivery to international buyers. FOB port facilities enhance supply chain reliability, while sample availability ensures confidence and transparency for trade partners. Choose Tenvir for prompt and professional logistics support in every shipment.
Commercial Application and Suitability of Tenvir Tablets
Tenvir Tablets are primarily indicated for the treatment of HIV-1 infection and chronic hepatitis B, making them highly valuable in clinical and hospital settings. Suitable for adult patients, these allopathic tablets may also be used in combination regimens depending on practitioner advice. Their pharmaceutical grade ensures reliable results, while the well-established safety profile allows wide applicability where prescription medication for viral infections like HIV is required.
Export Market Reach and Shipping Solutions for Tenvir Tablets
Tenvir Tablets have secured a formidable presence across global export markets due to their quality and unbeatable price point. Supplying from India, logistics are streamlined to provide express shipping and efficient delivery to international buyers. FOB port facilities enhance supply chain reliability, while sample availability ensures confidence and transparency for trade partners. Choose Tenvir for prompt and professional logistics support in every shipment.
FAQ's of Tenvir Tablets:
Q: How should Tenvir Tablets be stored to maintain their efficacy?
A: Tenvir Tablets should be stored below 30C and protected from moisture to maintain their quality throughout their shelf life of 2 to 3 years.Q: What conditions are Tenvir Tablets indicated for?
A: Tenvir Tablets are primarily used for treating HIV-1 infection and chronic hepatitis B as prescribed by a healthcare professional.Q: When can I expect the shipment of Tenvir Tablets after placing an order?
A: With express shipping options and efficient logistics, expect prompt delivery once your order is processed, depending on your export market location.Q: Where are Tenvir Tablets manufactured and supplied from?
A: Tenvir Tablets are manufactured and supplied directly from India, conforming to international pharmaceutical standards.Q: What are the common side effects associated with Tenvir Tablets?
A: Some individuals may experience nausea, headache, abdominal pain, or dizziness while using Tenvir Tablets.Q: What is the salt composition and dosage strength of Tenvir Tablets?
A: Each Tenvir Tablet contains 300 mg of Tenofovir Disoproxil Fumarate, conforming to high pharmaceutical quality standards.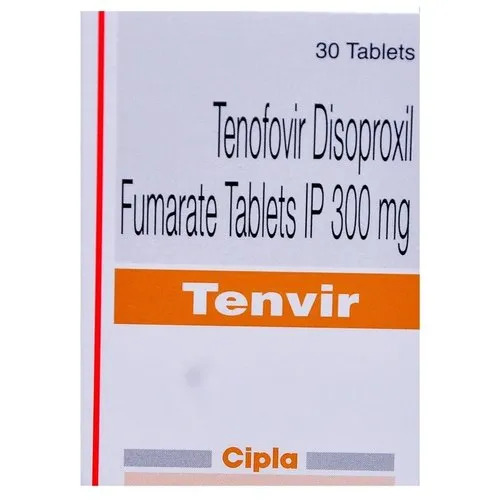
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in HIV Drugs Category
Emcure Spegra 30 Tablets
Price 2500 INR / Pack
Minimum Order Quantity : 100 Packs
Molecular Formula : Dolutegravir (C20H19F2N3O5), Emtricitabine (C8H10FN3O3S), Tenofovir Alafenamide (C21H29O5N6P)
Usage : For the treatment of HIV1 infection
Salt Composition : Dolutegravir + Emtricitabine + Tenofovir Alafenamide
Grade : Pharmaceutical Grade
Cipla Anti Hiv Drugs
Price 1200 INR / Bottle
Minimum Order Quantity : 100 Bottles
Molecular Formula : C21H30N6O4S
Usage : Used for the treatment of HIV infection
Salt Composition : Lamivudine & Zidovudine
Grade : Pharmaceutical Grade

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS